UCI Chem 131C Thermodynamics and Chemical Dynamics (Spring 2012)
Lec 06. Thermodynamics and Chemical Dynamics -- The Rotational Partition Function --
View the complete course: ../courses/chem_131c_thermodynamics_and_chemical_dynamics.html
Instructor: Reginald Penner, Ph.D.
License: Creative Commons BY-NC-SA
Terms of Use: ../info.
More courses at http://ocw.uci.edu
Description: In Chemistry 131C, students will study how to calculate macroscopic chemical properties of systems. This course will build on the microscopic understanding (Chemical Physics) to reinforce and expand your understanding of the basic thermo-chemistry concepts from General Chemistry (Physical Chemistry.) We then go on to study how chemical reaction rates are measured and calculated from molecular properties. Topics covered include: Energy, entropy, and the thermodynamic potentials; Chemical equilibrium; and Chemical kinetics. This video is part of a 27-lecture undergraduate-level course titled "Thermodynamics and Chemical Dynamics" taught at UC Irvine by Professor Reginald M. Penner.
Thermodynamics and Chemical Dynamics (Chem 131C) is part of OpenChem: ../openchem/
Recorded on April 13, 2012.
Index of Topics:
00:05 - Introduction
00:19 - Announcements
01:10 - What's in this Lecture?
01:25 - Neglecting electronic energy levels...
01:41 - Vibration, Rotation, Translation Diagrams
02:19 - but we're lucky. We can treat each of these energetic manifolds separately...
02:51 - ...consider first a monoatomic gas in one dimension...
03:43 - ...so its molecular partition function, q, is:
04:29 - Diagram: Calculation of Ground State and Excited State
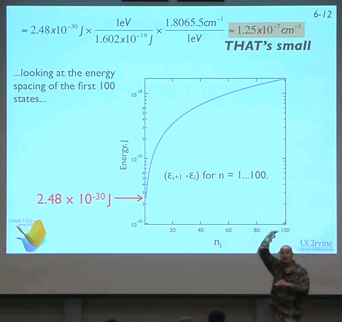
05:02 - Diagram: ...looking at the energy spacing of the first 100 states...
05:40 - ...If these states are quasi-continuous, we can rewrite this summation...
06:08 - Diagram: ...so after integration we have:
06:13 - Example: Calculate...
07:22 - Diagram: ...is related to the molecular properties through the mass...
08:28 - In terms of...
09:24 - Diagram: the enthalpy, H...
09:52 - Chart: so we can calculate everything for ideal, monoatomic gases...
11:10 - ...this begins to fulfill the promise of Statistical Mechanics:
11:41 - ...works for all ideal molecules.
11:51 - we have a manifold of rotational states that looks like this...
12:20 - ...and the energies of these states are given by the expression...
14:13 - ...B here has units of joules:
14:53 - ...I'm easily confused, so I try to stick to Joules:
15:26 - so let's work out the expression for...
16:01 - our usual expression for q applies for each of these three orthogonal axes:
16:27 - ...so if we write out this series, here's what it is:
18:36 - when B is expressed in Joules, these are the equations for the rotational partition function that apply:
19:43 - what's a symmetry number?
21:14 - huh? what axis?
21:59 - Ammonia
22:36 - Number of Symmetries:
26:55 - Example: Exercise 17.4a What is the symmetry of:
31:11 - Example: What is the symmetry number of:
Required attribution: Penner, Reginald Thermodynamics and Chemical Dynamics 131C (UCI OpenCourseWare: University of California, Irvine), ../courses/chem_131c_thermodynamics_and_chemical_dynamics.html. [Access date]. License: Creative Commons Attribution-ShareAlike 3.0 United States License.