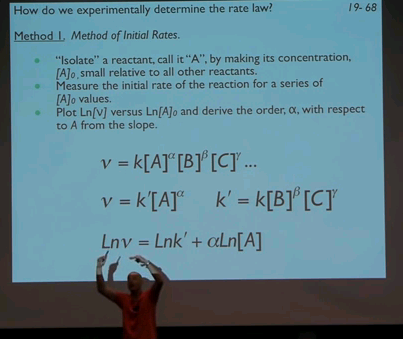UCI Chem 131C Thermodynamics and Chemical Dynamics (Spring 2012)
Lec 19. Thermodynamics and Chemical Dynamics -- Observational Chemical Kinetics --
View the complete course: ../courses/chem_131c_thermodynamics_and_chemical_dynamics.html
Instructor: Reginald Penner, Ph.D.
License: Creative Commons BY-NC-SA
Terms of Use: ../info.
More courses at http://ocw.uci.edu
Description: In Chemistry 131C, students will study how to calculate macroscopic chemical properties of systems. This course will build on the microscopic understanding (Chemical Physics) to reinforce and expand your understanding of the basic thermo-chemistry concepts from General Chemistry (Physical Chemistry.) We then go on to study how chemical reaction rates are measured and calculated from molecular properties. Topics covered include: Energy, entropy, and the thermodynamic potentials; Chemical equilibrium; and Chemical kinetics. This video is part of a 27-lecture undergraduate-level course titled "Thermodynamics and Chemical Dynamics" taught at UC Irvine by Professor Reginald M. Penner.
Thermodynamics and Chemical Dynamics (Chem 131C) is part of OpenChem: ../openchem/
Recorded on May 16, 2012.
Index of Topics:
00:06 - Introduction: Observational chemical kinetics
00:17 - Henry Louis Chatelier (1850 - 1936)
00:25 - Henry Lous Chatelier..."I let the discovery of the ammonia sythesis slip..."
02:21 - Le Chatelier's Principle says, for example, that with an increase in the total...
02:47 - example: Can we determine the relationship between...
03:06 - answer:
06:30 - what about the influence of temperature on K?
08:12 - That's it for Thermodynamics (Topic discussed in Chapter 17)
10:11 - Diagram: where are we?
12:30 - the first chemical subjected to kinetic analysis?
13:12 - some notation & jargon...a stoichiometric reaction...
17:30 - so an elementary reaction is one in which the indicated products...
17:53 - we discussed the extent of reaction...
18:52 - this is not as confusing as it looks. Here's an example...
20:19 - in terms of the extent of reaction...
22:44 - ...for this generation reaction...
23:20 - in terms of the extent of reaction...
23:30 - A rate law relates the concentration of reactants...
28:23 - for stoichiometric reactions, the rate law can not be deduced by inspection.
30:08 - Often, reactions are significantly reversible and both the forward and backward...
32:10 - be reminded that these simple expressions apply only because...
33:35 - what are the units of the rate constant in this case?
34:46 - Method 1. Method of Initial Rates
40:05 - Method 2. Use an integrated rate law.
43:59 - Method 2. (continued) Half-life
45:17 - Graph: (length of half of line is constant.)
45:55 - How do we experimentally determine the rate of law
Required attribution: Penner, Reginald Thermodynamics and Chemical Dynamics 131C (UCI OpenCourseWare: University of California, Irvine), ../courses/chem_131c_thermodynamics_and_chemical_dynamics.html. [Access date]. License: Creative Commons Attribution-ShareAlike 3.0 United States License.